
Unlike some medical device regulators, the New Zealand Medicines and Medical Devices Safety Authority (Medsafe) does not require pre-market approval or licensing for medical devices. Instead, the New Zealand medical device registration process involves notifying all medical devices through the Web-Assisted Notification of Devices (WAND) database before they can be marketed or sold. However, despite the absence of pre-market approval, Medsafe takes a rigorous and proactive approach to post-market monitoring of medical devices. Devices that do not perform safely according to their intended purpose can be recalled from the New Zealand market.
In this article, we will walk you through the notification process for medical devices in New Zealand and what you need to do to maintain compliance with MedSafe requirements long term.
An Overview of the New Zealand Medical Device Registration Process
Medical devices sold in New Zealand are regulated by Medsafe under The Medicines Act, 1981, which was revised in 1984 and 2003. The 2003 revision established the requirement to notify new devices in WAND. WAND functions purely as a database so that Medsafe can monitor device safety and notify Sponsors of recalls. There is no approval or license associated with WAND registration, and no fee is required. All devices sold in New Zealand must be entered in WAND within 30 days of retaining a Sponsor. Devices exempt from notification are listed in Schedule 1 of the Medicines (Database of Medical Devices) Regulations 2003.
Medsafe may request technical documentation for your device to verify its safety. Many manufacturers selling in New Zealand leverages European CE Marking, US FDA Clearance, or Australian TGA registration for this purpose
New Zealand Medical Device Registration Regarding IVDs
IVDs are considered exempt devices according to Schedule 1, though they can be notified in WAND on a voluntary basis. There is no recognized classification scheme for IVDs. However, IVDs are still obligated to comply with the Medicines Act, 1981, and its Regulations.
In-country Representation Requirements
A New Zealand Sponsor is the entity legally responsible for the device and obligated to ensure the device is safe when used for its intended purpose. An importer, exporter, or manufacturer can act as a Sponsor as long as they are located in New Zealand and operate a business there. Manufacturers without a legal entity in New Zealand must designate a Sponsor to notify their device in WAND, among other essential tasks:
-
- Updating the device information in WAND to reflect any device changes or correct inaccuracies within 10 days.
- Maintaining distribution records of supplied devices.
- Notify Medsafe of any device recalls or corrective actions.
- Ensure device labeling is consistent with GHTF guidance on labeling and Instructions for Use.
- Verify that all advertising for the device complies with the Medicines Regulations 1984.
Changing Sponsors requires the cooperation of both your new and incumbent Sponsors. The incumbent Sponsor must mark all your device notifications as obsolete in their WAND Sponsor account so the new Sponsor can resubmit your device notifications under their account.
New Zealand Medical Device Registration Regarding Classification
Your device classification does not dictate market entry requirements in New Zealand. Except for certain exempt devices, devices of all classifications require notification in WAND. However, device classification is required information for your WAND notification, along with the device’s Global Medical Device Nomenclature (GMDN) code.
New Zealand follows the Global Harmonization Task Force classification scheme. Devices are classified according to 22 rules found in Schedule 2 of the Medicines (Database of Medical Devices) Regulations 2003. If more than one rule applies to your device, the rule associated with the highest risk classification should be applied. Medsafe does not offer pre-market consultation regarding device classification, so Sponsors are responsible for identifying the correct risk classification
The classification scheme includes five classification tiers and two subclassifications:
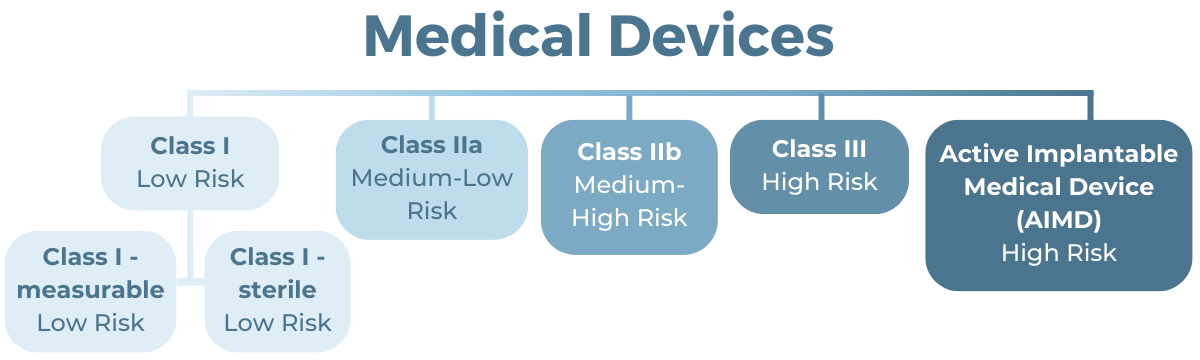
Some devices can be grouped into a single WAND notification as long as they are Class IIb or lower. To group devices, they must be made by the same manufacturer and share the same classification, GMDN code, intended purpose, and method of use.
How to Register your Medical Device in WAND
Only your Sponsor can notify your device as they will have sole access to WAND. Before beginning the device notification entry, your Sponsor will need the following information:
-
- Manufacturer name, address, contact information, and web address
- Device classification according to Schedule 2
- GMDN code
- Australian Register of Therapeutic Goods (ARTG) number if your device is registered in Australia (optional)
- Intended purpose
- Detailed device description
- Sterilization method if the device is sterile
The notification entry requires answers to a series of questions about the device’s characteristics. Each question will have a drop-down menu of selections. Medsafe may request additional information depending on your answers. Nevertheless, if the notification entry is complete when you submit and sign the compulsory declaration, WAND will confirm the new notification.
Device notifications should be updated by the Sponsor within 10 days of any changes. WAND is not a public-facing database; only your Sponsor and Medsafe staff can access the information about your device.
New Zealand Safety Monitoring for Medical Devices
Most medical device regulators impose an obligation on device manufacturers to proactively monitor their devices’ safety throughout the device life cycle and take initiative to prevent and correct safety issues. In New Zealand, Medsafe empowers healthcare providers, users, and patients to report device safety and quality concerns, as well as adverse events, directly to Medsafe.
All reports undergo a risk assessment by Medsafe staff to determine the severity, chance of recurrence, etc., to recommend the type of investigation appropriate for the report. Reports are assigned to an investigator who works directly with the Sponsor and device manufacturers to resolve the issue. Potential resolutions can include:
-
- Communications to providers and users via the Medsafe website and publications
- Continued monitoring
- Product modifications
- Revisions to the instructions for use or product labelling
- User education
In severe cases, Medsafe will recall the product from the market. They will also communicate safety information about your products to regulators in other jurisdictions, particularly the Australia TGA.
Ready to Start Registering Medical Devices in New Zealand?
New Zealand medical device registration is straightforward if your device’s safety is well established in other markets, and you have in-country representation through a Sponsor. MedEnvoy has personnel and operations in New Zealand ready to sponsor your devices. Contact us today to learn how we can assist with New Zealand WAND notification.





