
Harmonized standards in the European Union (EU) play a crucial role in ensuring that products, services, and processes comply with EU legislation. These standards are developed by recognized European Standards Organizations (ESOs)—such as CEN, CENELEC, and ETSI—following requests from the European Commission.
While the use of harmonized standards is voluntary, manufacturers, economic operators, and conformity assessment bodies that utilize them benefit from a “presumption of conformity” with relevant EU legislation. The European Commission has published eight new harmonized standards relevant to medical devices and IVDs:
- Six under the MDR (Implementing decision – EU – 2025/681 – EN – EUR-Lex)
- Two under the IVDR (Implementing decision – 2025/679 – EN – EUR-Lex)

Newly Harmonized Standards Under the IVDR
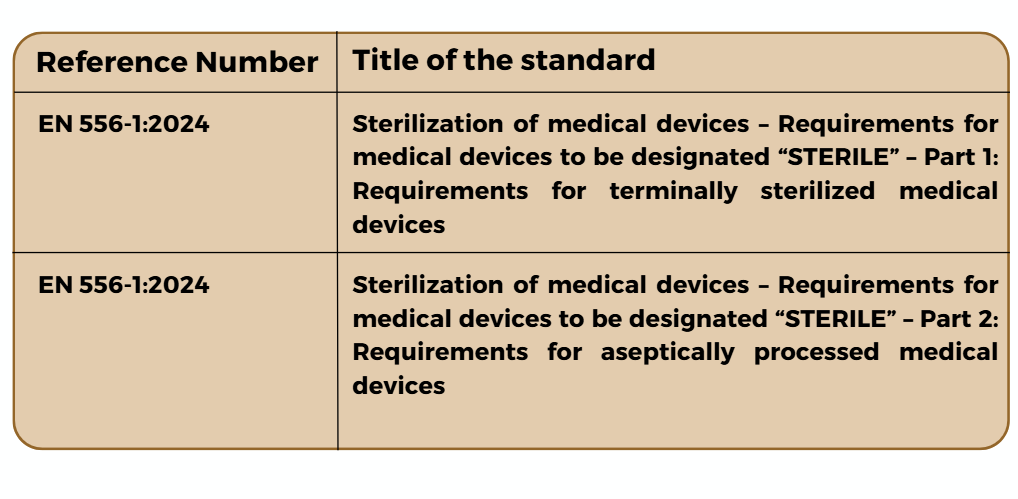
For more information, click here. For further information on how to take proactive steps for compliance with the updated standard, contact us.
____________________________________________


