
UDI Registration in Swissdamed: Key Insights and Deadlines
With economic operator registration in the Actor module of Swissmedic’s Swissdamed database in force for Swiss and Liechtenstein-based device manufacturers, authorized representatives, and importers, manufacturer attention has to turn to the Devices module. Unique device identification (UDI) registration in Swissdamed becomes mandatory from July 1, 2026 with a 6-month transition period up to December 31, 2026 except in cases of vigilance (i.e. reporting serious incidents, field safety corrective actions, or trends). It is recommended that manufacturers prepare for mandatory Swiss UDI registration. In this article we provide an overview of Swiss UDI and information regarding registration with Swissmedic likely to drive functional aspects of the device registration module of Swissdamed.
UDI in Switzerland and Liechstestein
Both the Swiss Medical Devices Ordinance (MedDO) and Ordinance on In Vitro Diagnostic Medical Devices (IvDO) establish UDI requirements under Article 17 and Article 16, respectively. These articles leverage the obligations and modalities associated with device identification under the EU MDR and IVDR, respectively, which include the requirement to assign a Basic UDI-DI to devices in addition to the device identifier (UDI-DI) and product identifier (UDI-PI). Additionally, under these articles:
- Manufacturers and system and procedure pack assemblers must assign to devices, systems or procedure packs, with the exception of custom-made devices, and all higher levels of packaging, a UDI prior to placing them on the Swiss or Liechtenstein markets.
- UDI must be stated on the labeling of the device, system or procedure pack and all higher levels of packaging (with shipping containers not considered to be a higher level of packaging).
- These economic operators must maintain an up-to-date list of all UDIs it has assigned as part of the technical documentation established to fulfill the requirements of Annex II to the MDR / IVDR.
- UDIs assigned by these economic operators must be registered in accordance with Article 29 of the MDR and Article 24 of the IVDR, respectively.
However, given that Switzerland is not part of the European Economic Area and the Mutual Recognition Agreement (MRA) between Switzerland and the EU has not been updated, EUDAMED registration is not capable of fulfilling the requirements for device registration in this market. Therefore, both the MedDO and IvDo include provisions regarding existing notification requirements which continue to remain in force in the absence of such functionality in the Swissdamed database.
UDI Swissdamed Registration in Switzerland
The Swissdamed device / UDI registration module is currently in development, with sections of this module anticipated for public release in 2025 for voluntary registration of certain devices until registration becomes mandatory.
In preparation for the rollout of the device / UDI registration module, two actions were undertaken by Swissmedic:
1. A test was performed in June 2024, with Swiss manufacturers performing mass uploaded of devices in a XML format. This test was successfully completed and allowed for Swissmedic to implement improvements to the Swissdamed platform.
2. A survey targeting organizations in charge of master data, supply chain managers or specialists from procurement and logistics for the purpose of gathering information on the use of master product data in Switzerland, to help guide priorities in the device / UDI registration module. While this survey of 451 companies revealed that the vast majority of UDIs are issued by GS1 (84%; n = 187), it also revealed important information on organizational capabilities and preferences for device / UDI data transmission methodologies with the following results:
Data Transmission Capability
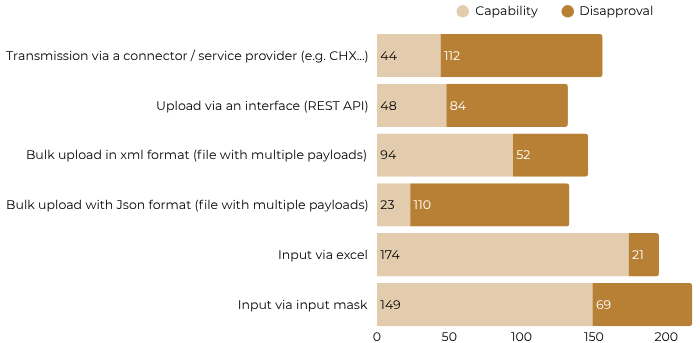
Source: Swissdamed Survey Results Report (23.01.2023 – 19.02.2023) available here.
Data Transmission Methodology Preference
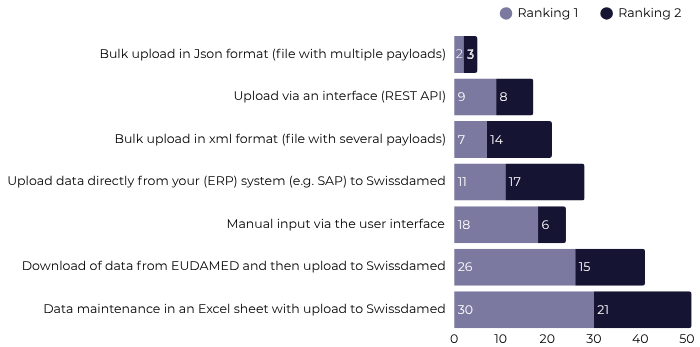
Source: Swissdamed Survey Results Report (23.01.2023 – 19.02.2023) available here.
75% of respondents indicated that they would be ready for Swissdamed device registration at the same time as EUDAMED. These results provide a strong indication that data transmission methods to be adopted by Swissmedic and integrated into Swissdamed are highly likely to include the uploading of Excel spreadsheets or EUDAMED data which should facilitate registration of those organizations with devices already registered in EUDAMED.
Learn More About the Swiss Medical Device Regulatory Framework with MedEnvoy
This article provides an overview of UDI requirements under the Swiss regulatory framework and information on device registration for Switzerland and Liechtenstein under the MedDO / IvDO.
MedEnvoy and Swissdamed Registration
MedEnvoy can support you in ensuring your UDI Swissdamed registration process is smooth, efficient, and compliant. With our deep regulatory expertise and technical solutions, we can handle your initial Swissdamed registrations and maintain them throughout the lifecycle of your devices. We also provide ongoing support to ensure you meet the evolving requirements of Swiss regulations. The “Devices” module in Swissdamed will go live in several phases. After the first release in 2025, it will be possible to register certain devices following XML files in EUDAMED format. By 1 July 2026, all devices must be registered in Swissdamed. Manufacturers or Authorized Representatives (ARs) have until 31 December 2026 to complete device registrations. If a serious incident, a Field Safety Corrective Action (FSCA), or trend needs to be reported to Swissmedic, immediate registration will be required starting from 1 July 2026. Our team specializes in managing Swissdamed registrations. We ensure that all required device information is accurately uploaded into the swissdamed system, given the timelines above. For more information on Swissdamed registrations, click here.
Furthermore, MedEnvoy offers Swiss authorized representative and Swiss importer services. Please reach out should you need assistance by clicking here and for information about our regulatory experts click here.


