The EC has decided to amend the MDR with regards to the assignment of Unique Device Identifiers (UDI) for Contact Lenses on the 10th of July 2023 by introducing Regulation 2023/2197. In this article, we assess the changes made and their impact on manufacturers.
Purpose and implications of Regulation 2023/2197
The Regulation is composed of 5 preambles and 2 Articles. In summary, the 5 preambles state that the purpose of Regulation 2023/2197 is to:
-
-
- Maintain the importance of the UDI-DI (as introduced in the MDR) for identification and traceability purposes; and remind that regardless of this amendment the UDI-DI remains a core element that Manufacturers must implement and provide within EUDAMED.
- However, it recognizes that contact lenses are found in many variants; therefore, if we follow the MDR’s current assignment process of UDI-DI’s then there would be a massive number of UDI-DIs that would be assigned to similar contact lenses, which would then overwhelm EUDAMED.
- Therefore, to avoid point (2) above, contact lenses that have the same clinical and design parameter combinations should be grouped under the same UDI-DI which will be identified as the “Master UDI-DI”.
-
In practice this means that for Standard and Made to Order contact lenses, one (1) Master UDI-DI shall be assigned to a group of contact lenses when the contact lenses in question have the same combination of design parameters, including at least the same base curve and diameter. A new Master UDI-DI shall be assigned whenever there is a change to the design parameters, including base curve and diameter, or there is a change as listed in section 3.9 of Annex VI, Part C of the MDR (which are: name or trade name, device version or model, labelled as single use, packaged sterile, need for sterilization before use, quantity of devices provided in a package, critical warnings or contra-indications: e.g. containing latex or DEHP).
Simplifying UDI-DI structure with the introduction of the ‘Master UDI-DI
It is our understanding that with the addition of a “Master UDI-DI,” manufacturers of contact lenses will now have for its groups:
-
- A Basic UDI-DI; and
- A Master UDI-DI.
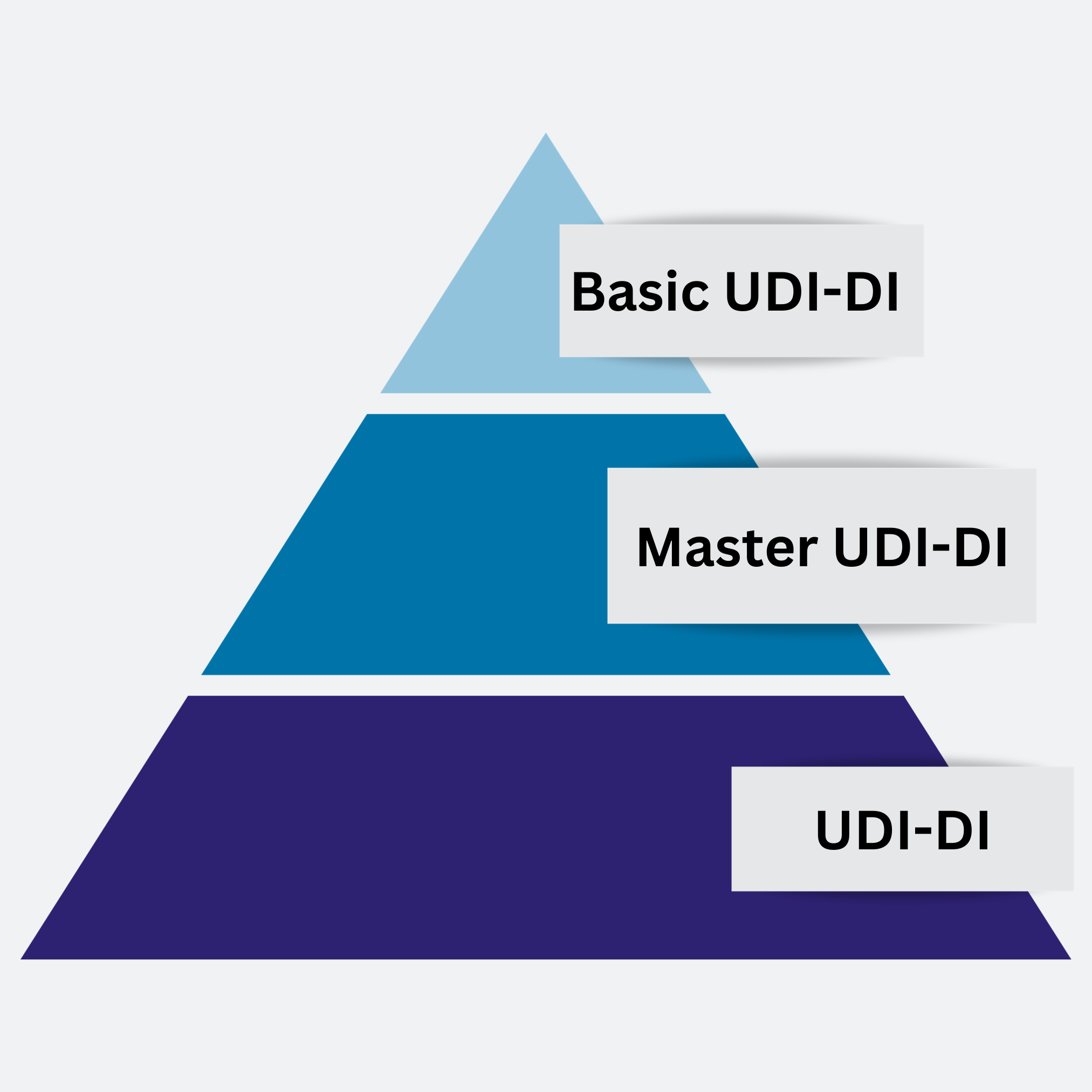
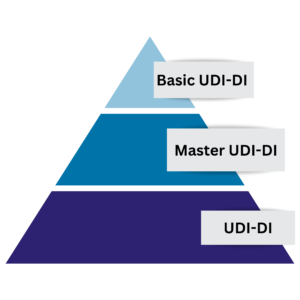
The concept of the Basic UDI-DI will remain the same and stays as described in the MDR; the idea of the “Master UDI-DI” is such that there will not be as many individual UDI-DIs anymore, instead they would be grouped such that there is one Master UDI-DI instead of, for example, 1000 UDI-DIs. Therefore, it would follow a “pyramid” concept where still the Basic UDI-DI is the overarching UDI (at the top), then there is a Master UDI-DI and lastly at the bottom there would be all the many product UDI-DIs. Mandating the inclusion of the Master UDI-DI for contact lenses is seen as an efficient measure to prevent EUDAMED from being inundated with an excessive amount of device data to upload.
Immediate impact of Regulation 2023/2197 on manufacturers of contact lenses
There is no immediate impact since the application date will be 9 November 2025. However, manufacturers are already allowed to assign a Master UDI-DI before that date.
Future impact of Regulation 2023/2197 on manufactures of contact lenses
From 9 November 2025, the changes introduced by this Regulation shall fully apply to manufacturers of contact lenses.
If you have any questions or require further insights on how Regulation 2023/2197 affects your operations, please don’t hesitate to contact us. Our team is ready to discuss how you can adapt to these changes and ensure compliance.


