
Inclusion of CH-REP in Communication on Vigilance Topics
Swissmedic has now specified that if the reporting is done by a manufacturer domiciled outside of Switzerland and Liechtenstein, the…
Stay informed with real-time regulatory news and expert insights, and keep the conversation going by connecting with us on social media.
Showing 217–225 of 229

Swissmedic has now specified that if the reporting is done by a manufacturer domiciled outside of Switzerland and Liechtenstein, the…
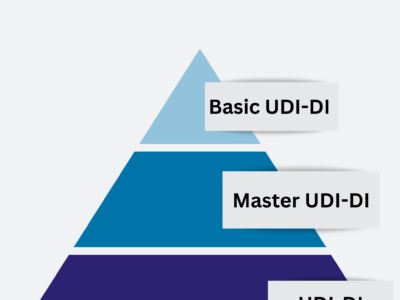
The EC has decided to amend the MDR with regards to the assignment of Unique Device Identifiers (UDI) for Contact…


As of August 31, 2023, there are changes to how adverse incidents involving medical devices should be reported to…

Due to the status of the agreement on mutual recognition of conformity assessments (MRA) between Switzerland and the…

The Dutch Health and Youth Care Inspectorate (the Inspectorate) suggests manufacturers be more aware of medical device incident reporting. In…

Virtual manufacturers of medical devices that do not need UK Approved Body/EU Notified Body approval are still required…

The UK Government has introduced the Medical Devices (Amendment) (Great Britain) Regulations 2023, allowing CE marked…

The UK government has made changes to the UK MDR (2002) to extend the acceptance of CE…
Showing 217–225 of 229