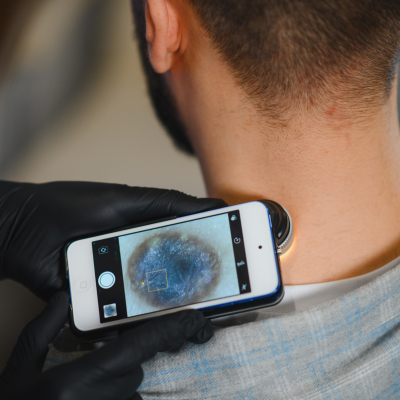
The Food and Drug Administration (FDA) issued a final order on March 25, 2026 reclassifying two types of skin-cancer-related diagnostic devices: (1) optical diagnostic devices for melanoma detection and (2) electrical impedance spectrometers, from Class III (high risk) to Class II (moderate risk).
These devices are now regulated as “software‑aided adjunctive diagnostic devices for use on skin lesions by physicians trained in the diagnosis and management of skin cancer.”
Key Points
- Reclassification & Regulatory Pathway
- The FDA determined that sufficient scientific evidence now exists to regulate these devices under Class II with special controls, which still ensure safety and effectiveness but reduce regulatory burden for manufacturers.
- These devices now follow the 510(k) pathway, not Pre-Market Approval (PMA), significantly reducing submission burden and review timelines.
- New Device Type Definition
- Devices are now categorized as: “Software-aided adjunctive diagnostic devices for use on skin lesions by physicians trained in the diagnosis and management of skin cancer.”
- For prescription use only and intended as adjunctive tools used after a dermatologist identifies a suspicious lesion.
- Not to be used as standalone diagnostic tools or to confirm a diagnosis.
- Special Controls Required
- To mitigate risks (e.g., false positives/negatives, device misuse, hardware/software failures), the FDA established comprehensive requirements, including:
- Clinical validation demonstrating >90% sensitivity for high‑risk lesions (or strong justification if lower), improvement in clinicians’ diagnostic accuracy compared to unaided assessment, and inclusion of risk factor across diverse patient populations, including skin phototypes, body sites, and risk factors.
- Human factors evaluation required to confirm safe and correct use by trained clinicians.
- Biocompatibility, sterilization, reprocessing validation required for patient-contact components.
- Labeling requirements and non-clinical testing (e.g. software verification/validation, electromagnetic, electrical, mechanical, thermal safety, hardware compatibility, device precision)
- Post‑market surveillance studies, unless FDA determines premarket data are fully sufficient.
- Scope Limitations
- The reclassification does NOT apply to mobile apps or layperson‑use tools. Only applies to clinician‑use, adjunctive diagnostic devices.
- Effective Date
- The final order took effect beginning April 24, 2026.
Impact on Manufacturers
- Transition from PMA → 510(k) pathway: Shorter review timelines, lower evidence burden, reduced compliance obligations (no PMA supplements or annual reports).
- Significant changes (especially to Artificial Intelligence (AI) / Machine Learning (ML) algorithms or hardware) will still require a new 510(k), unless included in a Predetermined Change Control Plan (PCCP).
- Reclassification expected to encourage innovation and improve access.
How MedEnvoy Can Support
MedEnvoy supports medical device manufacturers in navigating evolving FDA guidance by translating regulatory expectations into clear, actionable strategies. Our team provides end-to-end support across regulatory planning, FDA engagement, and clinical evidence development to help reduce uncertainty and avoid downstream delays. We assist with early FDA interactions, including premarket strategy and submission preparation. If you have any questions regarding this summary or the accompanying guidance document, we encourage you to reach out to our regulatory experts, here.