The publication of this Q&A is intended to assist manufacturers and other economic operators in understanding the impact of Regulation (EU) 2024/1860, which has amended (EU) 2017/746 (IVDR) with an extended transition period for applicable IVDs.
To learn more about how you can streamline your medical device to the EU market, click here.
Key Points for Regulation (EU) 2024/1860:
1. The extended transition periods only apply to “Legacy Devices”
-
- 31 December 2027, for class D devices;
- 31 December 2028, for class C devices;
- 31 December 2029, for class B devices and for class A devices placed on the market in sterile condition.
2. If a manufacturer has a “Legacy Device” and they do not wish to purse certification under the IVDR, they may continue to place the “Legacy Device” on the market until 26 May 2025 at a minimum, with extended transition periods possible if:
-
- “Legacy Device” has an IVDD certificate valid until 26 May 2025 AND (i) the devices do not present an unacceptable risk to the health or safety and (ii) the “Legacy Device” continues to comply with the IVDD (98/79/EC).
- “Legacy Device” is “self-declared” under the IVDD, and the manufacturer has implemented a quality management system (QMS) per IVDR Article 10(8) before 26 May 2025, they can remain on the market until 26 May 2025 (Class D), 26 May 2026 (Class C) or 26 <ay 2027 (Class B and Class A sterile).
Note: Refer to #4 below for additional information.
3. Manufacturers should have evidence that their “Legacy Device” benefits from the extended transition period, whether that is in the form of a “self-declaration” memo addressing the requirements of IVDR Article 110, or a “confirmation letter” or similar from their Notified Body.
4. To benefit from the extended transition period the manufacturer must lodge a formal application for conformity assessment with their notified body by:
-
- 26 May 2025 for devices with an IVDD certificate and for Class D devices, which were “self-declared” per the IVDD
- 26 May 2026 for Class C devices that were “self-declared” per the IVDD
- 26 May 2027 for Class B and Class A Sterile devices, which were “self-declared” per the IVDD
5. The manufacturer must have a written agreement in place with their Notified Body, as it relates to the IVDR certification by:
-
- 26 September 2025 for devices with an IVDD certificate and for Class D devices, which were “self-declared” per the IVDD
- 26 September 2026 for Class C devices that were “self-declared” per the IVDD
- 26 September 2027 for Class B and Class A Sterile devices, which were “self-declared” per the IVDD
6. Manufacturers must implement a quality management system (QMS) per IVDR Article 10(8), and the manufacturer must submit an application for review of the QMS per the timelines associated with the IVDR formal application, listed above.
Key Reminder:
The transitional periods described within the IVDR and the amendments to the IVDR, e.g., (EU) 2024/1860, do not apply to Class A devices. These devices are required to be fully compliant with the IVDR as of 26 May 2022. The only exception is for those Class A devices that are Sterile, for which a Notified Body is required, and thus the extended transition period.
Included in the Q&A is a Flowchart to assist manufacturers, and other economic operators, in determining whether their device is covered by the extended transitional period provided for in Article 110 of Regulation (EU) 2017/746 on in vitro diagnostic medical devices (IVDR), as amended by Regulation 2024/1860. Manufacturers are strongly encouraged to review the Flowchart in parallel with the IVDR and recently release regulations, e.g., (EU) 2024/1860:
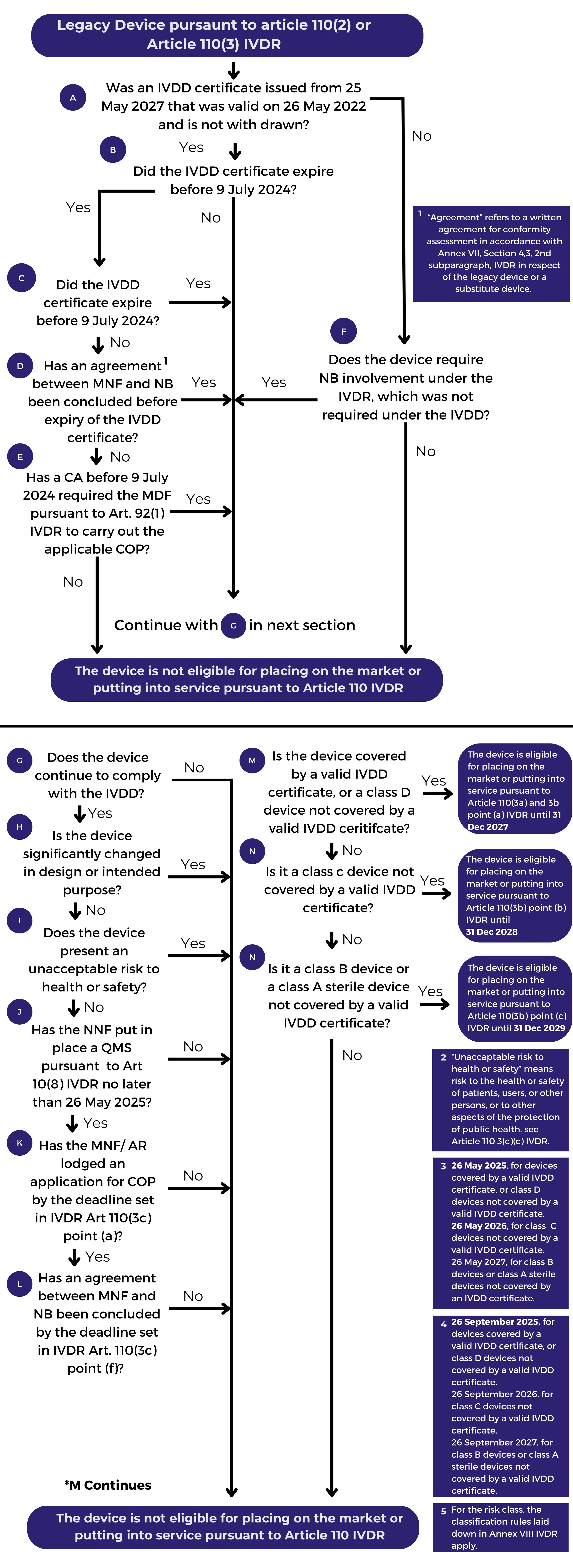
Notes:
“Legacy Devices” are those that are placed on the market or put into service after the IVDR’s date of application (i.e. 26 May 2022) if certain conditions are fulfilled. Those devices can be:
-
- Devices covered by a valid EC certificate issued by a notified body in accordance with Directive 98/79/EC on in vitro diagnostic medical devices (IVDD) prior to 26 May 2022;
- Devices for which a declaration of conformity was drawn up prior to 26 May 2022 in accordance with the IVDD and for which the conformity assessment procedure pursuant to the IVDR (contrary to the IVDD) requires the involvement of a notified body.
_____________________________________________
If you need help in understanding Regulation (EU) 2024/1860 for IVDs, contact us so our regulatory team can help you. To learn more specifically about our EU service, click here.


