
New MHRA Guidance on Clinical Investigations Notifications
On 27 August 2025, the UK’s Medicines and Healthcare products Regulatory Agency (MHRA) released an updated set of flow…
Stay informed with real-time regulatory news and expert insights, and keep the conversation going by connecting with us on social media.
Showing 1–9 of 11

On 27 August 2025, the UK’s Medicines and Healthcare products Regulatory Agency (MHRA) released an updated set of flow…

The United Kingdom Medicines and Healthcare products Regulatory Agency (MHRA), under the jurisdiction of the UK Department of Health and…
In this article, we share the advantages and disadvantages associated with consolidating the roles of authorized representative and importer.
As we’ve noted in a previous article, under the EU Medical Device Regulation (MDR) 2017/745 and…

For medical device and IVD manufacturers selling in the UK, much has changed in the last few years. First, the obvious: the UK is no longer part of the European Union.

If you export devices to the European Union, United Kingdom or Switzerland, you will need to appoint an “Importer of Record.” Understandably, many...
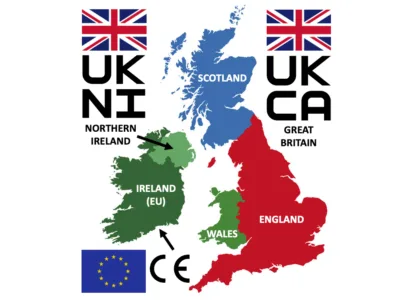
Ever since the United Kingdom (UK) left the European Union (EU), there have been lingering questions among medical device manufacturers about how the new UK regulatory

Pre-Brexit, the United Kingdom was a member of European Union (EU). As such they were obligated to transpose the EU Medical...

If you have been selling your medical devices or IVDs in Europe for some time, you’re probably aware of the requirements for identifying your EU Authorized...
Showing 1–9 of 11